Stability is essential to the human body. Millions of chemical reactions occur within our cells every second to sustain our lives, to aid organ work, to generate energy, and to repair tissues. To ensure the effectiveness of these reactions, the inner environment of the body should be under close control. PH regulation, especially in blood, is one of the most significant components of such an internal balance. The role of the pH balance in cellular functioning cannot be overestimated since even minor changes beyond its normal levels may cause an enzyme to be compromised, disrupt the entire organ system, and put a person at risk.
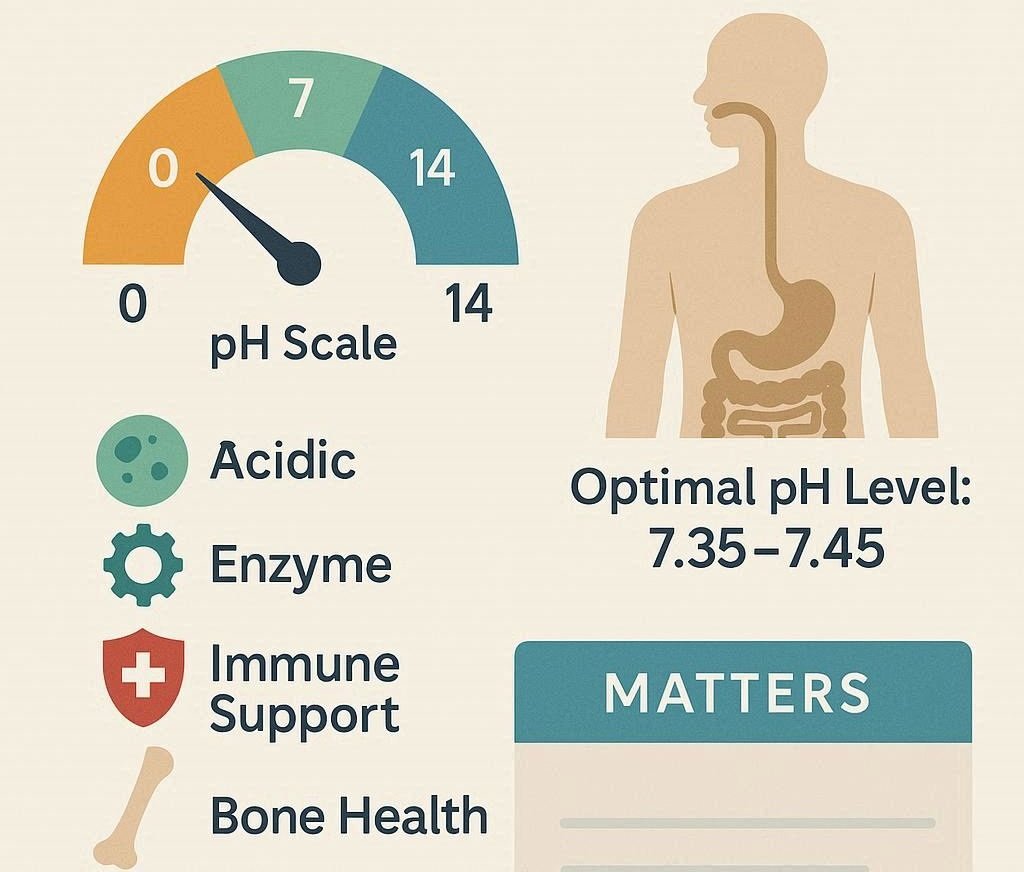
PH is a term that is used to describe the degree of acidity or alkalinity of a solution. It is rated on a scale between 0 and 14 with below 7 being acidic, above 7 being alkaline and 7 being neutral. The normal human body range of blood pH is very close; it is between 7.35 and 7.45 which is slightly alkaline. This is a small range that might not seem significant, yet it is of utmost importance. Even a few decimal points can change the capacities of proteins and enzymes to become effective, to change the delivery of oxygen and to disrupt the communication among cells.
This delicate balance is maintained in the body via a complex procedure of buffering systems, lungs and the kidneys. The combination of these mechanisms makes sure that acids and bases are in balance even though the production of the metabolic waste is constant. In this paper, the authors discuss the significance of pH balance in cellular activities, the biochemical mechanisms that regulate it, and the reasons as to why the process is so important to life.
Learning about pH and Why the Body Keeps pH at such a stringent Level
pH is a scientific unit that indicates the amount of hydrogen ions in a fluid. The denser the solution is, the more it is acidic. The less concentrated it is, the alkaline it is. Since our body comprises mostly of water-based fluids, pH is crucial in ensuring a stable internal environment.
The significance of pH balance in cellular activity is that almost all the biological processes rely on proteins and enzymes. Enzymes are special proteins which accelerate chemical reactions in the body. These proteins are optimally active in very narrow pH ranges. When the pH is altered excessively, enzymes can be out of shape and will work less efficiently.
An example is energy production in cells, muscle contraction, hormone signalling, nerve transmission, all of which depend on pH-sensitive enzymes. That is why the body should maintain the blood pH in the such range.
Even minor imbalances may lead to fatigue, confusion, weakness, difficulties in breathing and in critical cases organ failure.
The Significance of pH Homeostasis in Cellular Functions
To understand the significance of the pH balance in the functioning of cells, a brief examination of the activity inside the cells would help. All the body cells depend on a stable chemical environment to exist and carry out its functions.
Cells are composed of enzymes that help in breaking down nutrients, the production of ATP (energy), protein synthesis, and the elimination of waste products. Such responses are based on a constant pH atmosphere. At excessive acidity of the internal pH, proteins can start to denature, i.e. lose their three-dimensional form.
When proteins lose their shape, the proteins might not work properly anymore. This has the ability to influence cell membranes, receptors, transport channels, and intracellular communication pathways.
Another reason why pH balance is necessary is transport of oxygen. The red blood cell protein that carries oxygen is hemoglobin, which is pH sensitive. When the blood is excessively acid, hemoglobin will lose oxygen in a different way, potentially leading to a lack of oxygen in the tissue.
This shows that the pH balance has a direct effect on cellular activity and the reason why the body maintains it so rigidly.
Buffering Systems: Line of Defense
Buffering systems are the quickest and initial defense mechanisms of the body to changes in pH. Buffers are chemical compounds, which do not allow the pH to change abruptly due to the neutralization of excessive acids or bases.
The bicarbonate buffer system is the most significant buffer of the blood.
The mechanism of this system is the equilibrium of carbonic acid and bicarbonate ions:
CO2+H2O↔H2CO3↔H++HCO3−
This response enables the body to take up excess hydrogen ions rapidly when the acidity increases and to give out the hydrogen ions when the alkalinity rises.
This reaction is reversible and hence it gives quick short-term control of blood pH.
Phosphate buffers, and protein buffers (particularly hemoglobin) are other buffer systems.
These mechanisms assist in preserving the significance of pH balance to cellular activity by averting abrupt harmful changes.
Lung role in pH Balance
The lungs are also significant in the maintenance of blood pH since they control the level of carbon dioxide in the body.
Carbon dioxide is not a waste gas. As it dissolves in blood, it forms carbonic acid which influences pH.
CO2+H2O→H2CO3
In the event that the level of carbon dioxide is high, then acidity is high. When the amount of carbon dioxide decreases, blood becomes less acidic.
That is why the rate of breathing has a direct impact on pH
In case the blood becomes excessively acidic, the brain will notify the lungs to breathe faster. Increased breathing speed eliminates greater amounts of carbon dioxide, decreasing acidity.
- Once the blood is excessively alkaline, the breathing rate might decelerate a bit to preserve carbon dioxide.
- This respiratory regulation occurs in few minutes, and lungs are one of the rapid pH regulators in the body.
The health of respiratory is thus directly associated with the importance of pH balance in cellular functioning.
Kidney roles in the long-term pH regulation
The pH balance is regulated long-term by the kidneys, whereas the lungs regulate this balance in a short term.
- Kidneys keep the balance of acid and base through excretion of hydrogen ions and the reabsorption of bicarbonate.
- Due to the excess acidity of blood, the kidneys release more hydrogen ions into the urine and store bicarbonate.
Alternatively, when the blood is too alkaline, then they can secrete bicarbonate.
This is not as quick as breathing into the lungs, and can require hours or days, yet is necessary to maintain steady pH levels.
The long-term significance of pH balance to cellular functioning is thus seen to be centrally dependent on the kidneys.
What Occurs When the pH is out of Range
- Any minor changes in normal blood pH can be disastrous.
- When blood is excessively acidic the phenomenon is known as acidosis.
- When it gets excessively alkaline, then it is referred to as alkalosis.
- Both disorders can disrupt the activities of enzymes, nerve impulses, and muscles.
- The effects of acidosis can include fatigue, confusion, headache, rapid breathing and weakness.
- Alkalosis can result in muscle spasms, light-headedness, lack of sensation, and heart arrhythmias.
This indicates why the relevance of pH balance in regard to cellular functioning is directly related to survival.
The importance of pH Balance to Survival
Stringently regulated biochemical responses are vital to the existence of the body.
- Lack of stable PH causes failure of enzymes, alterations in oxygen delivery and inefficiency of cellular metabolism.
- Stable pH is needed by the nervous system, cardiovascular system, muscles and kidneys.
This is the reason why even minor changes are considered medical emergencies.
Regarding the significance of pH balance in cellular functioning, the significance of the balanced internal environment of the body is ultimately reflected.
Factors of the environment and health which influence pH
- Body pH balance can be affected by a number of factors.
- Carbon dioxide removal may be affected by respiratory diseases.
- Acid excretion might be reduced by kidney disease.
- PH may also be altered by dehydration, serious infections, diabetes, and long-term vomiting.
These circumstances indicate the precariousness of pH balance.
Conclusion
One of the most vital concepts in human physiology is the importance of pH balance for cellular function. The body maintains the blood pH in a narrow range of life sustaining range through the combined efforts of the buffering systems, lungs, and the kidneys.
This strict control prevents the damage of enzymes, helps deliver oxygen and makes sure that cells will be able to keep operating effectively.
In the absence of this balance, life sustaining processes would occur at a very high speed and hence pH control is key to general survival.