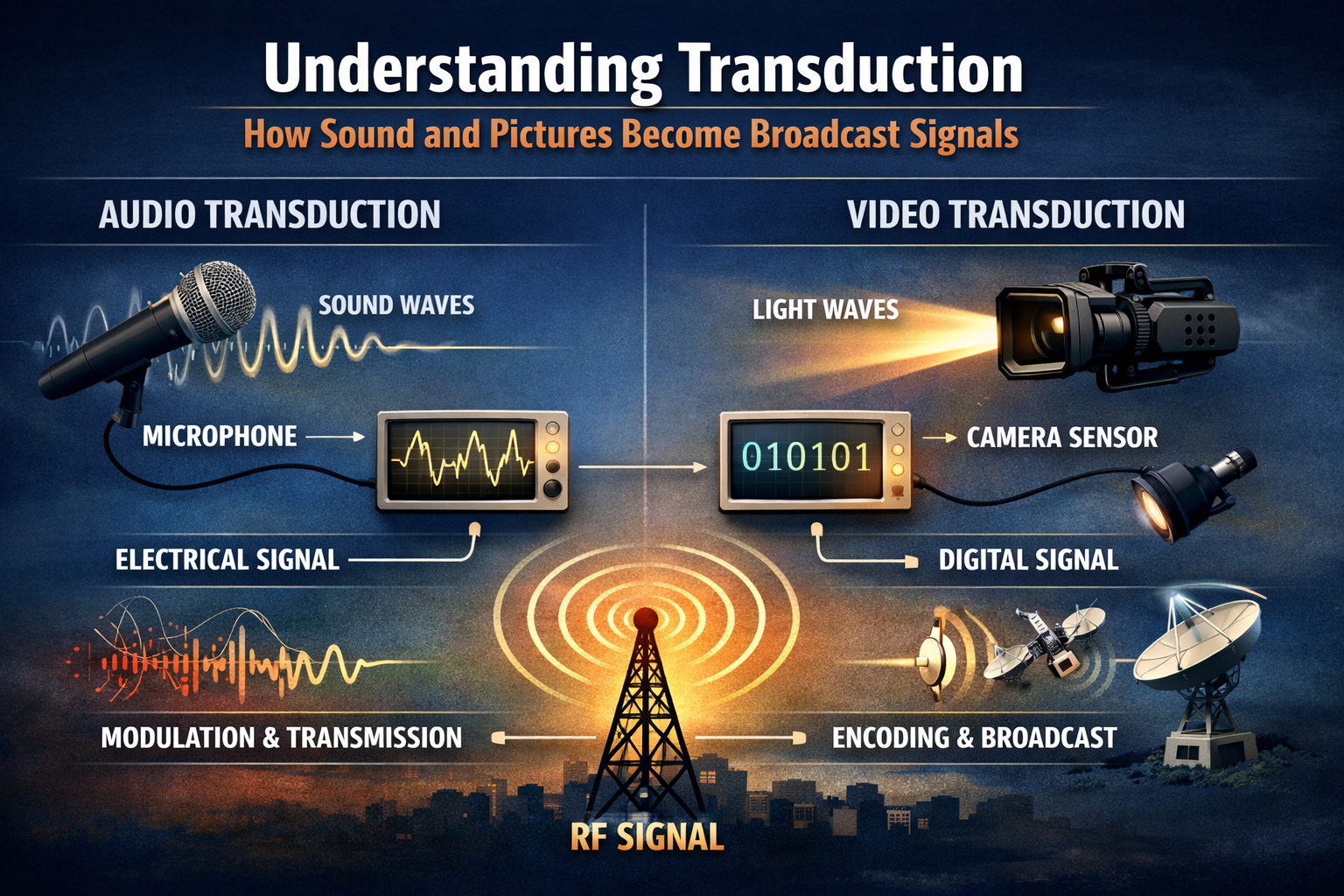
Water is one of the most essential substances on Earth, found in our bodies and in the oceans, rivers, and lakes. Water is also important for survival and has interesting properties. One example is the density of water. This information is valuable to scientists, engineers, and students.
In this guide, you will learn the basics of information relating to the density of water and the different aspects that accompany this phenomenon. This includes the reasons water density changes with temperature, methods for measuring and calculating water density in different units, and the practical uses of this information in day-to-day life. For students learning physics or chemistry, teachers preparing to teach, and anyone interested in the behavior of matter, this guide will be very useful.
Table of Contents
Complete Guide to the Density of Water with Formulas & Units
Common Units for the Density of Water
Density of Water Lbs per Gallon
What Affects the Density of Water
Practical Applications of the Density of Water
Common Misconceptions About Water Density
Why Students Learn About the Density of Water
How Online Tutoring Helps Students Understand Water Density
Easy Ways to Visualise Density
Final Overview of the Density of Water
What Is the Density of Water?
Water density is another way to describe the chemical and physical properties of water. It is a way of showing how much ‘matter’ is concentrated within a given ‘space’. The relationship between mass and volume can be used to calculate this.
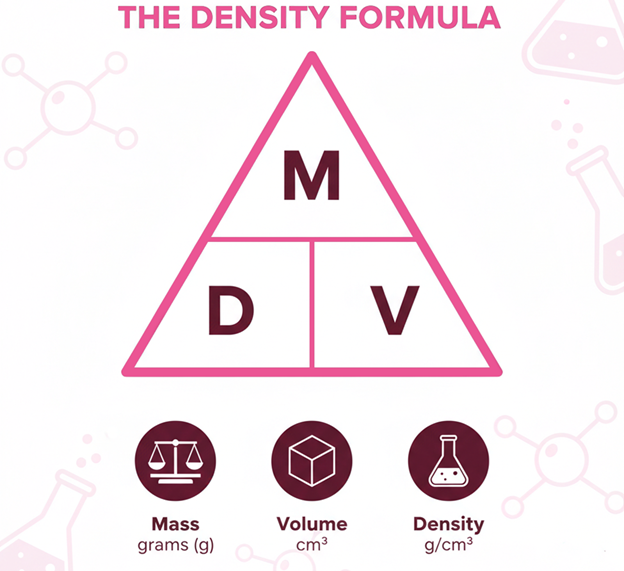
Density Defined
Simply put,
Density = Mass ÷ Volume
In simple words, the density of water is the measure of how much water weighs for the amount of space it occupies. Scientists determine the water density in the following fields:
- grams per cubic centimetre (g/cm³)
- kilograms per cubic meter (kg/m³)
- grams per millilitre (g/mL)
- pounds per gallon (lbs/gal)
- pounds per cubic foot (lb/ft³)
- kilonewtons per cubic meter (kN/m³)
Fields such as chemistry, engineering, and environmental science may use different units that represent different densities of water.
Common Units for the Density of Water
Here are some ways to state the density of water.
Density of Water in g/cm³
Grams per cubic centimetre is the most common unit used in the sciences.
- At approximately 4 °C: density of water in g cm³ ≈ 1 g/cm³
The above means that each cubic centimetre of water weighs approximately 1 gram.
Density of Water in g/mL
Grams per millilitre is also another common form:
- density of water g ml ≈ 1 g/mL
This is also the same case, as 1 mL is equivalent to 1 cm³.
Density of Water in kg/m³
In metric physics or engineering:
- density of water in kg/m³ ≈ 1000 kg/m³
This means one cubic meter of water has a mass of 1000 kilograms.
Density of Water in lb/ft³
In U.S. customary systems:
- density of water in lb/ft3 ≈ 62.4 lb/ft³
This figure is used in construction and fluid mechanics.
Density of Water in kN/m³
An engineering-friendly but rare unit is:
- density of water in kN/m3 ≈ 9.81 kN/m³
This approximates density to weight under Earth’s gravity.
Density of Water in g/L
When larger liquids are measured:
- density of water in g l ≈ 1000 g/L
This is equal to 1 kg per litre.
Density of Water Lbs per Gallon
In casual everyday American volume terms:
- density of water lbs gallon ≈ 8.34 lbs/gal
Hence, one U.S gallon of water weighs 8.34 pounds.
Why All These Units Matter
An academic discipline or profession may require a specific unit of measure among the ones provided:
| Unit | Typical Use | Example Question |
| g/cm³ | Chemistry labs | What is the density of water molecules? |
| g/mL | Biology or food science | How heavy is water in a test tube? |
| kg/m³ | Physics & engineering | How much does water weigh per cubic meter? |
| lb/ft³ | Civil engineering | How much does water weigh inside a foundation? |
| lbs/gal | Everyday measurement | How much does a bucket of water weigh? |
| kN/m³ | Structural engineering | What is the weight force of water? |
Being able to convert between various units of measurement is useful when solving different practical problems. This may include understanding buoyancy or even building a plumbing system.
What Affects the Density of Water
Water’s density varies under different conditions. The following are the main contributing factors.
Temperature
Water’s density varies with temperature.
- At 4 °C, water’s density is at its maximum, which is 1 g/cm³.
- At temperatures above and below 4 °C, water’s density is lower.
Water molecules behave unusually: they expand when warmed and form ice crystals when cooled.
Impurities and Salinity
These values apply only to pure water. When other materials dissolve in water, such as salt, the density increases. As a result, seawater is denser than freshwater, which is why ocean water floats differently from river water.
Pressure
Under normal circumstances, the effect of pressure is minimal. However, in deep oceans or laboratory environments, pressure alters molecular configurations only slightly.
Density of Water Formula
The formula for density is as follows:
Density = Mass / Volume
The formula can be used with any system of measurement for mass and volume, as long as they are in the same system, either metric or U.S. customary.
1000 grams and 1000 cubic centimetres of water are examples of this.
- Density = 1000 g / 1000 cm³ = 1 g/cm³.
For water, density is 1 g/cm³. Density can also be referenced as 1000 kg/m³ or 62.4 lb/ft³ for engineering purposes.
Water Density and Temperature
Water density varies with temperature. As the temperature drops, the density of water increases. This explains why ice floats and why bodies of water separate (stratify) in winter.
| Temperature (°C) | Density (kg/m³) | Approx. (g/cm³) |
| 0 (near freezing) | ~999.8 | ~0.9998 |
| 4 (maximum density) | 1000 | 1 |
| 20 | ~998.2 | ~0.9982 |
| 25 | ~997.0 | ~0.9970 |
| 100 (boiling) | ~958.4 | ~0.9584 |
Practical Applications of the Density of Water
Knowing the density of water is useful for many reasons.

Buoyancy and Floating
An object can float if its density is lower than that of water.
- Due to the lower density of water when compared to the average density of a ship, it can float.
- The same goes for a balloon: the air inside is less dense than water.
This is one of the more important principles that are taught in engineering or physics design.
Shipping and Maritime Science
One of the most important factors that affects how cargo ships are designed and built is density. It is vital for the ship’s operational and functional elements, such as:
- Load limits
- Safety considerations
- Port depth requirements
Climate Science
Density is responsible for creating the oceans’ most powerful currents. Salt water is denser than fresh water, and cold water is denser than warm water. These variations of temperature and salinity vertically distribute the water, creating circulation patterns and zones of current flow. Understanding density is vital in the study of circulation patterns.
Lab Measurements
In the lab, water density is used as a reference standard when:
- calibrating hydrometers
- calculating concentrations of solutions
- studying chemical reactions.
Fun Facts About Water Density
Ice Floats!
Water is unique among other liquids in that it expands when it freezes.
Why do lakes freeze from the top down? Water is less dense than ice. Because of this, the ice layer on a frozen lake keeps the water beneath it from freezing. This keeps aquatic life alive during winter.
Water at 4 °C Is Special.
Water has unique behavior when it is at 4 °C. At this temperature, water is at its densest point. Water’s odd behavior at this temperature helps to stabilise bodies of water when the seasons change.
Common Misconceptions About Water Density
- Ice is heavier than water, a common mistake. In fact, ice is less dense than water, and that’s why it floats.
- Water density is constant; this is also false. Water density varies due to temperature, pressure, and the presence of other substances.
- Unit conversion is optional, incorrect! Unit conversion is vital in calculations; converting from g/cm³ to lb/ft³ is necessary.
Why Students Learn About the Density of Water
While studying the three basic sciences of physics, chemistry and earth sciences, density is an important basic concept as it describes:
- The relationship of mass, volume and how matter behaves
- The explanation of various natural phenomena
- The approach to problem-solving and the interpretation of data
- The concept is measurement-based, reinforcing lab skills
Students who have difficulty with the concept of density or with units like density of water lbs /gallon) or density of water (g/mL) can get professional help on the Mixt Academy platform. They offer one-on-one online tutoring sessions designed for students’ schedules.
How Online Tutoring Helps Students Understand Water Density?
When learning about water density, students often struggle with unit conversions, formulas, and apps. Online tutoring centres like Mixt Academy help students understand the concepts of physics and chemistry. Students can benefit from:
✔ One-on-one assistance with physics and science
✔ Abstract the application of the density of water formula
✔ Learn the conversion between unit ranges of kg/m³, lb/ft³, and g/cm³
✔ Understand related exam questions
✔ With examples, gain the confidence to use real and practical problems.
Common Mistakes Students Make
When learning about the density of water, students tend to struggle with:
- Mixing units like grams and kilograms
- Not remembering the temperature alters the density
- Confusing weight with density
- Not knowing how to operate the formula
- Incorrectly seeing the units with the density of water in ft or in mg/L
Expert online tutoring can fix most of these confusions before the exams.
Easy Ways to Visualise Density
Water vs Oil
Oil is less dense than water, so it floats.
Rock in Water
A rock sinks because it is denser than water.
Bottle Experiment
Fill a bottle with water, measure its weight and volume, and calculate the density using the simple formula.
Converting Density Units
Let’s look at some basic conversion examples:
Convert g/cm³ to kg/m³
Since: 1 g/cm³ = 1000 kg/m³
If water is 1 g/cm³:
- 1 g/cm³ × 1000 = 1000 kg/m³
Convert kg/m³ to lb/ft³
1 kg/m³ ≈ 0.0624 lb/ft³
So:
- 1000 kg/m³ × 0.0624 ≈ 62.4 lb/ft³
Convert lb/gal to kg/m³
1 lb/gal ≈ 119.83 kg/m³
So:
- 8.34 lb/gal × 119.83 ≈ 1000 kg/m³
To compare results across fields, it is necessary to convert units.
Final Overview of the Density of Water
When learning about water density, understanding the behaviour of matter, the effects of temperature on matter, and applying units to real-world problems are important skills to consider. Important Points:
- The temperature of 4 °C (degrees Celsius) is where water has maximum density.
- The units g/cm³, kg/m³, lb/ft³, and lb/gal are examples of units used to measure the same quantity.
- The density of a substance is affected by temperature and impurities.
- Density is calculated using the formula Density = Mass ÷ Volume.
A solid understanding of density is important, whether it be to use in the conversion of the density of water into foot (ft) units, understanding the patterns in a graph, or solving a scientific problem.
FAQs
What is the density of water at room temperature?
At 20°C, the density of water is approximately 998kg/m³ or 0.998 g/cm³.
Is the density of water 1 g/ml?
A common unit of water density is gram per millilitre (g/ml) or gram per cubic centimetre (g/cm³). Actually, the exact density of water is not exactly 1 g/ml, but rather slightly less, at 0.9998395 g/ml at 4.0° Celsius (39.2° Fahrenheit).
Why does water expand when it freezes?
When water freezes to ice, it expands. The molecules in the water take a crystalline shape, and that’s why ice is less dense than water and floats.
How do you determine the density of water in pounds per gallon (lbs/gal)?
One U.S. gallon of water is approximately 8.34 lbs. Therefore, the weight of the water gives the water’s density in lb/gal and describes the relationship between mass and volume in U.S. customary units.
Is the density of water 1000 or 1?
The density of water is taken as 1000 kg/. The value of the density of water (i.e., 1000 kg/ ) signifies that there is 1000 kg of water present in 1 volume of water.
Does the density of water in grams per millilitre (g/mL) change with temperature?
Yes, at 4 °C, the density of water is almost 1 g/mL; it is slightly less than 1 g/mL at higher temperatures.
Can density be used to identify a substance?
Yes, if you consider that density is a unique characteristic of a substance, you can determine the density of an unknown sample by comparing it to a known sample, such as water.
Is the density of water 1 kg/m³?
Relation to other measures. The density of water is about 1000 kg/m³ or 1 g/cm³, because the size of the gram was originally based on the mass of a cubic centimetre of water.
How can students get help with water density problems?
Students struggling with formulas, unit conversions, and water density labs can get expert online tutoring from Mixt Academy.
What is the density of water at 4 C?
The density of water at 4°C (standard temperature) is 1 g/cm³ or 1000 kg/m³. The SI unit of density is kilogram per cubic metre (kg/m³).