The term CRISPR has turned to be not only a buzz term within the scientific community, but in the mainstream media and popular discussions. However, behind such a revolutionary creation as the gene-editing tool, there is an extraordinary story of origin the beginning of which does not lie in the realm of high technologies-graded laboratories but rather in a tiny, unnoticeable world of bacteria. The article follows the history of CRISPR technology since its emergence as an example of bacterial immunity to being one of the most striking solutions in genetic engineering.
Anchor Link: Find out more about using the modern CRISPR technology and its history of transforming the medical field and agriculture sector.
The CRISPR.
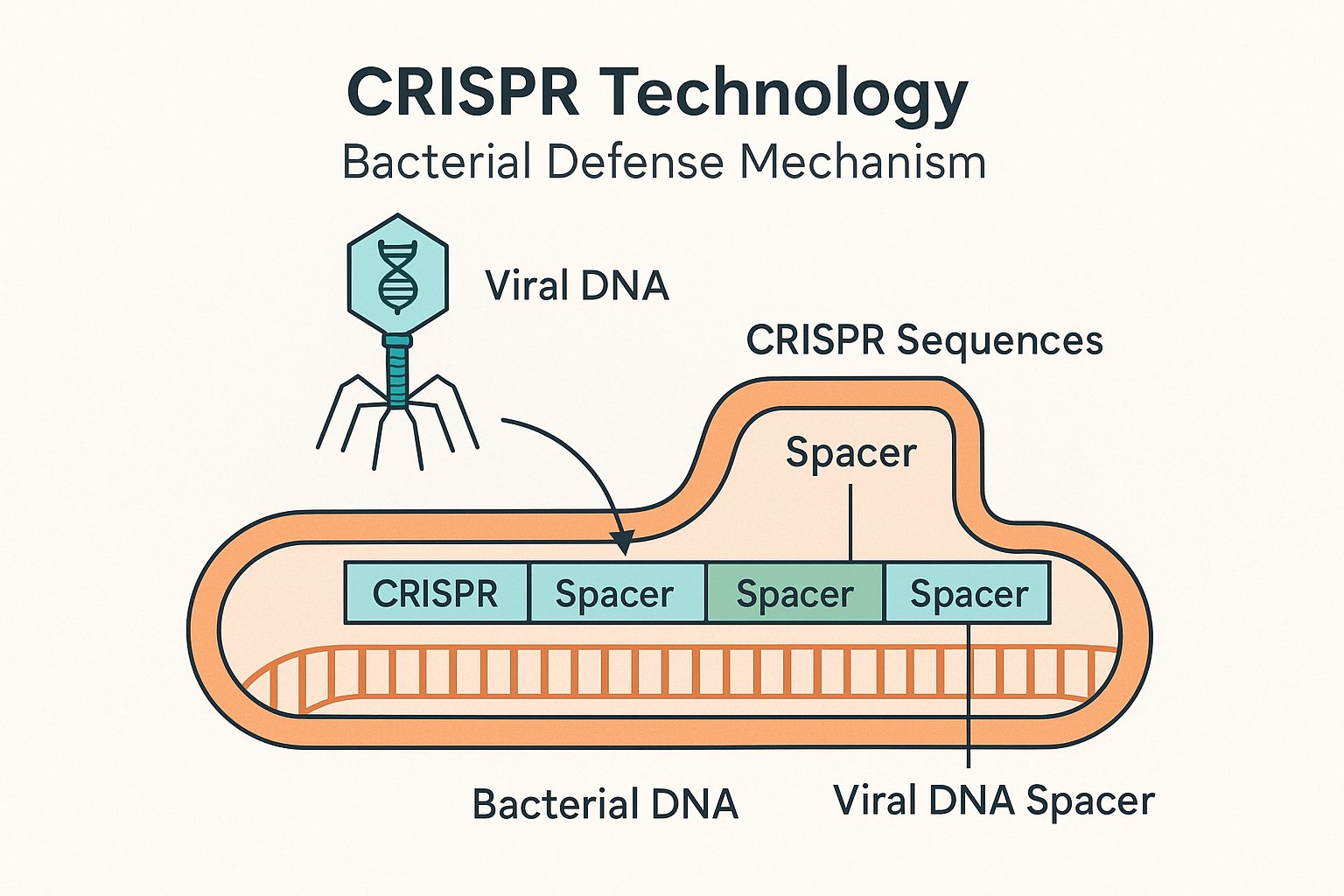
The CRISPR refers to the Clustered Regularly, Interspaced Short Palindromic Repeats. These compositions are particular strains of DNA present in the genome of bacteria and archaea. Although it might seem to be a technical term, the role of the acronym is rather obvious, it serves as a sort of a biological memory, which can aid bacteria in combating viruses.
Just as human beings, bacteria are prone to attacks by viruses. They can store the snatches of the DNA of the virus that hits them in a form of inserting it into their genome, when they are attacked by those viruses known as bacteriophages. The CRISPR region has these DNA segments and stores them. By the second attempt that the same virus moves to invade, the bacterium can identify the virus and develop quick strike against it using special proteins known as the Cas proteins but especially Cas9.
The First Glimpses: CRISPR into the Bacterial DNA
The history of the CRISPR started in an unobtrusive way in the end of 1980s. The unusual repeating DNA sequence with spacers in between were discovered in 1987, when Japanese scientist Yoshizumi Ishino and his cadre were working on E. coli. The relevance of these sequences was then quite ununderstood. The patterns later were detected in numerous other bacteria and archaea, but they went ignored as genetic junk.
The puzzle began to click in to place only when a Spanish microbiologist called Francisco Mojica, at the University of Alicante, started to look at these sequences in detail in the 1990s. Mojica observed that the spacer regions between repeat sequences had similarity with viral DNA. He supposed that these sequences were one kind of defense applied by bacteria–a memory of a former genetically stimulated incidence of a virus.
This was a rather daring thought, which was not instantly accepted by the rest of scientific world. However, it is through the work of Mojica that was bringing results slowly and precisely that what turned out to be one of the most revolutionary concepts in the field of biology developed
CRISPR-Cas: The Microbial immune system
In the early 2000s researchers started to confirm the hypothesis of Mojica. They found that CRISPR sequences were linked with other CRISPR (Cas) genes coding to enzymes capable of cleaving DNA. The most famous of them is Cas9 that is comparable to molecular scissors that can be used to cut DNA at specific sites.
The CRISPR-Cas system acts in fact in three steps:
- Adaptation: Bacteria cells engulf viral-DNA fragments and follow them up in their CRISPR arrays.
- Expression: Such arrays are copied into RNA molecules, to direct such enzymes Cas.
- Interference: In case of the same virus once again, the RNA leads the Cas proteins to the DNA of the invader and cuts it down by destroying it.
This process is amazingly like contemporary gene-editing systems. The leap of science was, however, still to be made.
The Turning Point: CRISPR to the Matter of Gene Editing
It was in 2012 when the actual breakthrough was made possible by Emmanuelle Charpentier and Jennifer Doudna. Developing the microbial model of immune defense, they demonstrated that the CRISPR-Cas9 system could be redirected to cut the DNA in any arbitrary position-not only in bacteria, but also in plants, animals, and human cells.
Scientists now had a method of making Cas9 cut specific genes by developing custom RNA guides to point the way to them. What this implied was that:
Silence of defect genes
• Additional genes insertion
• Mutation The restoration of naturally occurring mutations.
By such a revolutionary breakthrough, Charpentier and Doudna became the recipients of the 2020 Nobel Prize in Chemistry.
Their contribution made CRISPR into a programmable, effective, and cheap gene editing tool that opened up a new world in the medical, agricultural and many more fields.
The Fast Growth of CRISPR: five Milestones
The scientific community soon understood the huge potential of CRISPR and the key milestones were reached:
2013 – Human cells First use
Around the beginning of 2013 several research groups showed that CRISPR-Cas9 could be used to edit the human genome with exquisite specificity in a cell. This concept of evidence marked the beginning of another generation of biomedical research.
2015 Two rivers brought gene editing to living animals using CRISPR.
Replies revealed that CRISPR can edit the genes in live animals such as mice and zebrafish, paving the way to disease modeling and possible treatments.
2016- Pilot study in humans
The first study of human clinical trial was introduced by China to treat lung cancer using CRISPR. Shortly afterward the West was looking into the application of CRISPR in managing sickle cell disease and some blindness.
2020- The Nobel Prize Awards
The final whistle was blown by the Nobel prize given to Doudna and Charpentier showing the CRISPR is the heart of genetic future.
CRISPR: It is Not Just a Lab Thing
In the modern times, the CRISPR does not remain a technology of academic laboratories. It is moulding:
- Medicine: Therapy against genetic diseases such as sickle cell anemia, cystic fibrosis and also some cases of cancer.
- Agriculture: Creation and development of disease resistant crops, increasing yield, and decreasing the use of pesticides.
- Conservation: The possibility of restoring extinct or dying in nature due to DNA editing of the close relatives.
Serial entrepreneurs, biotech behemoths and universities are spending big on CRISPR technology to develop therapies to treat diseases previously considered untreatable.
Future Possibilities and Ethical Question
The common opinion about CRISPR is connected with the rapid development of this technology, which causes the increased world debate. There is general agreement on therapeutic application of the use in the consenting adults; however, when it comes to editing the human embryo or germlines it is a very serious ethically questionable matter.
In 2018, a Chinese scientist He Jiankui shocked the world by claiming to have created two genome-edited twin girls based on the CRISPR method. The news evoked outrage and demands of greater regulation across the world. The scientists of the world are currently joining forces with ethicists and policymakers with the aim of putting real boundary lines.
In the meantime, new versions of CRISPR, such as base editing and prime editing, provide even greater control to minimize unintended consequences and increase the variety of the diseases that could be addressed.
The Significance of the origin of CRISPR
It is not only an academic exercise to learn about the origins of CRISPR as a part of bacterial immune defense, but the ability to be interested in something is evidence of the strength of a curiosity-based research. The scientists that initially observed strange discordateness in bacteria never imagined that they have landed a tool that may one day cure genetic diseases.
The story of this obscure corner in the field of genetics of microbes and a major innovation, awarded by the Nobel Prize scientific knowledge is exemplified and shows how unpredictable, and at the same time capable making a great difference, science is.
That science, what is a mystery today can become a miracle tomorrow, in the medical development.
Final Thoughts
The story of CRISPR begins on the salty shores of Spanish marshes where Mojica used to gather his archaea and continues into high technology labs where human genes are rewritten in order to create a fascinating story of discovery, risk, and promise. An ancient defense mechanism in bacteria now is transforming medicine, agriculture and our own perception of life.
The background story as we proceed to experiment further and get deeper into the field of gene editing when we recollect, it reminds us of the beauty of science not only about its results, but also about the process.